Glucose is the main energy source in all living cells, regardless if those cells are idly growing in a petri dish in a lab, in the leaf of your household plant, or one of the billions networked within the human body.
In animals, cell get energy by directly eating foods with sugar, or by digesting more complex starches, such as the carbohydrates found in pasta and rice, into the simpler sugar, glucose, which is then easily absorbed into the bloodstream. But how the body handles those sugars – and how many sugars you bombard yourself with – can have wide ranging implications.
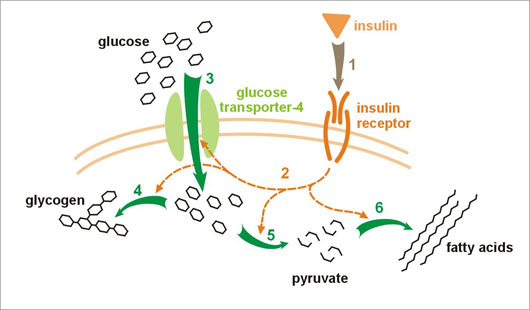
The process is more complicated than you might think, as shown in the figure below. So let’s take a closer look at exactly how glucose gets into cells – and how that could eventually lead to alzheimers.
Understanding Glucose
After chowing down on your favorite meal, glucose is literally coursing through your veins. Being a small molecule, glucose is rapidly shuttled around the body. It makes its way out of our blood vessels, and moves into the tissues of our body, primed to deliver energy to hungry cells. But despite its ambition, it can’t cross the cell membrane to get to the inside of cells where it’s converted to energy. Special proteins, called glucose transporters, line cell membranes and help pass glucose from the outside to the inside of cells. There are several different types of glucose transporters found throughout in the body — for instance, GLUT-1 is found in erythrocytes (red blood cells) and the endothelial cells that line blood vessels. GLUT-3 is found in neurons. GLUT-4 is found in fat and muscle cells.
Most glucose transporters simply simply act as a conduit, allowing glucose to passively move through the cell membrane. But GLUT-4, the transporter found in fat and muscle cells, is rather unique, because it must be activated by the hormone, insulin. The process goes something like this: the pancreas, sensing the glucose rush in the bloodstream after a meal, sends the hormone insulin to the rescue. When near a muscle cell, insulin binds to a membrane protein called an insulin receptor. This binding sends a chemical message to the GLUT-4 protein that it’s time to start letting glucose in.
Most cells don’t require a constant source of glucose. Rather, they can store an energy reserve as glycogen, giving us a backup boost of fuel after we’ve burned through all the available glucose. You’ve inevitably heard the expression “hitting the wall” during a distance running competition, like a marathon. Physiologically, “hitting the wall” means you’ve burned through all the glucose AND glycogen in your body, and now you’ve got to convert fat to energy, which is a much less efficient process, and leads to extreme fatigue.
Diabetes
Now that we have a basic understanding of how glucose acts in the body, let’s shift our attention to what can go wrong with this pathway.
Diabetes, a disease affecting millions of people, occurs when something goes wrong with the insulin-glucose interaction in cells. In Type I diabetes, there is simply not enough insulin circulating in the bloodstream. Tissues that have GLUT-4 transporters, which require insulin to get glucose into cells, starve.
In Type II diabetes, cells have become insulin-resistant, or “insensitive” to insulin. This means that there is plenty of insulin in the bloodstream to go around, but either the hormone can’t bind to the insulin receptor, or once bound, there is no chemical message sent from the insulin receptor to the glucose transporter to start allowing glucose to enter. The pancreas, sensing there is still a large amount of glucose in the bloodstream, releases more and more insulin, compounding the situation.
In both Type I and Type II diabetes, the high level of glucose in the blood can damage organs such as the heart, the kidneys, or the eyes, just to name a few. But as we’ve previously discussed, an increase in glucose circulating in the blood also means that there is an elevated level of insulin meandering through our body. Although insulin’s primary function is to signal to GLUT-4 transporters that it’s time to move some glucose across the cell membrane, insulin is a hormone with far-reaching biological effects. Let’s see what happens to my organ of choice, the brain, when levels of glucose and insulin skyrocket.
Sugar and the Brain
Now let’s talk about how energy is supplied to neurons, because it is a bit different than most cells in the body.
Approximately 20% of our resting metabolic rate goes to power our brains. This means our brains require more energy at rest than all of the muscles in our body, or our heart and kidneys combined.
Energy in the brain must be supplied by a constant source of glucose, since limited stored energy in the form of glycogen exists. When glucose levels in the blood drop too low, neurons will eventually stop functioning, and brain damage occurs.
Given that insulin is required for glucose uptake in other parts of the body, early studies sought to determine if insulin had any effect in the brain. In 1979, researchers showed that the level of insulin in the brain determined how much food a group of monkeys consumed, but they could not produce a direct link to how insulin worked in the brain.
Later studies found large amounts of both the GLUT-1 transporter, which carries glucose from the blood steam to brain tissue, and the GLUT-3 transporter, which moves glucose into neurons. Since both the GLUT-1 and the GLUT-3 do not need insulin to initiate the process of moving glucose from one place to another, we prematurely concluded that glucose metabolism in the brain has nothing to do with insulin.
It turns out that our initial thoughts were way off. Fast-forward to present day, where we now know that neurons need insulin to survive. We’ve actually identified several areas of the brain with neurons that have insulin receptors. Two particular areas with insulin receptors, the cerebral cortex and hippocampus, are heavily involved in memory formation and organization.
The hippocampus also has GLUT-4 insulin-dependent transporters, which are shuffled around to maximize glucose absorption in neurons when an animal is learning a new task. Other studies have shown that glucose metabolism in the cortex is at least partially controlled by insulin. Injection of insulin caused an increase in glucose uptake in the cortex, presumably due to either activation of GLUT-4 transporters, or by insulin stimulation of neurons that increased the need for cellular fuel.
Is There a Link Between Diabetes and Alzheimer’s Disease?
In Alzheimer’s Disease, funky, abnormal protein clumps collect in the brain. Physicians and researchers call these protein clumps neurofibrillary tangles or amyloid-beta plaques, depending on whether they’re actually inside cells, or in the space between neurons. Whether the proteins themselves are the cause or the result of the disease is still unclear. Regardless, we know that Alzheimer’s Disease causes neurons to die, which leads to cognitive problems, such as memory issues and confusion.

Alzheimer’s Victim
Given the finding that glucose metabolism in the brain is not entirely independent of insulin, and the surge of Type II diabetes, scientists have developed an interest in how sugar acts on on brain diseases. There are two lines of thinking when it comes to the link between Alzheimer’s Disease and diabetes. The first says that the elevated levels of insulin, as seen in Type II diabetes, may cause Alzheimer’s Disease. Studies point to insulin in causing neurofibrillary tangles to form inside of cells, as well as affecting the processing of the amyloid precursor protein, which form those senile plaques in between cells.
The second theory of Alzheimer’s Disease and diabetes says that Alzheimer’s Disease is not caused by the increase in insulin from Type II diabetes. Rather, there is improper insulin handling occurring directly in the brain. People are calling this ‘Type III Diabetes’.
A group at Northwestern University, led by neurobiologist William Klein, is leading the research behind this new potential form of diabetes that has its sights set on the brain. The Northwestern group believes that small peptides, called amyloid-beta derived diffusable ligands (ADDLs), break away from senile plaques, and attack cells. These ADDLs have been shown to remove insulin receptors from the brain, thereby impairing the synapse, which is the area where neurons connect to each other. Klein’s group showed that certain drugs, which increased insulin sensitivity, eliminated the harmful effects of ADDLs on neurons, which according to Klein shows that ”bolstering insulin signaling can protect neurons from harm“.
So Now What?
For years, sugar has been the silent killer, slowly chipping away at our body’s sensitivity to insulin, and contributing to the obesity epidemic. We still don’t have enough evidence to say for sure whether there is a direct link between Alzheimer’s Disease and diabetes. But the fact that glucose- and insulin-signaling irregularities cause problems in the brain creates yet another reason to restrict the sugar and refined carbs in our diets.